Abstract
Targeted therapies emerged over the past decades as a fundamental component of the anti-cancer therapeutic arsenal. Although leading to a significant improvement in patients' (pts) outcome, their use is often associated with development of resistance mechanisms driven by cancer cells adaptability or clonal selection of pre-existing resistant subclones.
The clinical use of JAK inhibitors like ruxolitinib (rux) was introduced a decade ago for treatment (trt) of myeloproliferative neoplasms (MPN) driven by JAK/STAT pathway activation. Here, we seek to evaluate the effect of JAK inhibitors on MPN clonal evolution and its impact on clinical outcome.
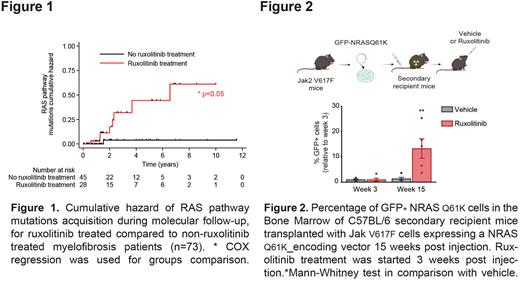
First, we interrogated a cohort of 73 myelofibrosis pts for which longitudinal molecular evaluation was available (NGS panel: 36 genes). We analyzed molecular evolution prior to and after rux trt, on all pts for which rux was initiated between 2 molecular evaluations (n=22). After a median exposure of 27 months IQR[15; 70], 16 patients acquired a total of 35 mutations. 40% of these mutations occurred within genes involved in the RAS signaling pathway (NRAS, KRAS, CBL). Rux was independently associated with RAS mutations acquisition (HR 9.8 CI95%[1.2; 78.9], p=0.031, COX model) (Figure 1).
Next, we investigated whether rux positively selects RAS mutant clones through ex-vivo trt of primary CD34+ hematopoietic cells derived from RAS mutated MPN pts (n=4). RAS mutations allelic frequency increased across all 4 patients upon rux trt. Single cell DNA sequencing was then applied to these samples to study their clonal architecture. RAS mutations were either present within the MPN driver clone (harboring a JAK2, CALR or MPL mutation), or in a clone without driver mutation, suggesting that rux-induced RAS clonal selection can occur both in a JAK/STAT hyper-activated or wild-type (WT) context. Furthermore, longitudinal molecular evaluation of 2 of these pts treated with rux for their MPN showed an increase in the allelic frequency of RAS mutations over time, confirming in vivo the impact of rux trt on RAS mutations selection.
Interestingly, whereas the presence of RAS mutations did not influence clinical outcome of MPN pts not treated with rux, such mutations were in contrast, independently associated with poor clinical outcome; decreased transformation free (HR 6.7 CI95%[1.9; 23.2], p=0.003) and overall (HR 3.1 CI95%[1.3; 7.1], p=0.008) survivals in rux treated pts.
The effect of JAK inhibition on RAS clonal selection was further validated using JAK2WT and JAK2V617F mutated cell lines and murine models. First, a competition assay between cells overexpressing an NRASQ61K plasmid and control cells showed an increased cellular fitness of NRASQ61K cells upon rux trt, in HEL and UKE-1 JAK2V617F mutated cell lines. This effect was rescued by trametinib trt confirming its dependence on the MAPK pathway activation. In addition, overexpression of NRASQ61K within JAK2V617F (HEL, UKE-1) and JAK2WT (MV4-11, U937) cell lines rescued rux-induced cellular viability decrease in vitro. Rux also increased the in vitro colony forming ability of NRASQ61K overexpressing primary murine cKIT+ hematopoietic cells and gave them a competitive advantage in vivo in a JAK2V617F driven mouse model (Figure 2). Similarly, rux trt resulted in increased cellular fitness of NRASG12V hematopoietic cells in an in vivo competition assay.
Finally, we confirmed that JAK2 suppression using shRNAs increased NRASQ61K mutated cells fitness in cellular competition, viability and colony forming in vitro assays, thereby corroborating the direct effect of rux-induced JAK inhibition on RAS clonal selection. Mechanistically, this effect resulted from a decreased RAS-induced senescence as evaluated by β-galactosidase staining in HEL and UKE-1 cell lines.
Overall, our study reveals an unanticipated selection of RAS mutations upon rux exposure, negatively impacting clinical outcomes. Careful screening for pre-existing RAS mutated clones before rux trt, and the search for alternative therapies for pts with RAS mutated MPN could be proposed to avoid such deleterious clones' expansion.
From a more fundamental perspective, the enhanced oncogenic potential of RAS mutations after inactivation of the oncogenic JAK/STAT pathway, pinpoints an intriguing paradoxical oncogenic mechanism and highlights the complexity of the combinatorial mutational landscape in cancer.
NM BR and JJK BC LB: equal contribution
Disclosures
Soret:Novartis: Consultancy. Gou:Rigel Pharmaceuticals: Other: supplies fostamatinib for our NIH funded clinical trials . Vainchenker:Incyte: Research Funding. Padua:VivaVacs Ltd: Current Employment. Kiladjian:Incyte: Membership on an entity's Board of Directors or advisory committees; AbbVie: Membership on an entity's Board of Directors or advisory committees; Novartis: Membership on an entity's Board of Directors or advisory committees; AOP Orphan: Membership on an entity's Board of Directors or advisory committees; BMS: Membership on an entity's Board of Directors or advisory committees. Benajiba:Gilead: Research Funding; Pfizer: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal